Mannitol: More Than Just a Sugar Substitute
Historical Development
Mannitol’s story stretches way back, before the days of shiny pharmaceutical labs and high-speed manufacturing lines. Early records show this sweet compound turning up in the sap of certain trees and plants, tapped by ancient cultures who recognized its value even without knowing its chemistry. In the 19th century, European chemists isolated it during studies on plant sugars and named it based on the manna-like exudates that dripped from ash trees. Years later, as chemical processes advanced, factories ramped up production to meet growing interest in sugar alternatives and new ways to treat medical conditions.
Product Overview
Walk into a pharmacy, step into a bakery, or tour a medical facility — mannitol shows up in more places than most folks realize. Often sold as a white, crystalline powder or free-flowing granules, this sweetener has a straightforward taste profile without aftertastes, carving out a spot on ingredient lists for people needing low-calorie or sugar-free options. In medicine, doctors rely on high-purity mannitol to shift body fluids or draw water away from swollen tissues, which shows just how far this little sugar alcohol has come. Big players in food production, pharmaceutical manufacturing, and technical industries buy it in bulk, bagged and labeled with specific quality grades to match safety standards set by regulators.
Physical & Chemical Properties
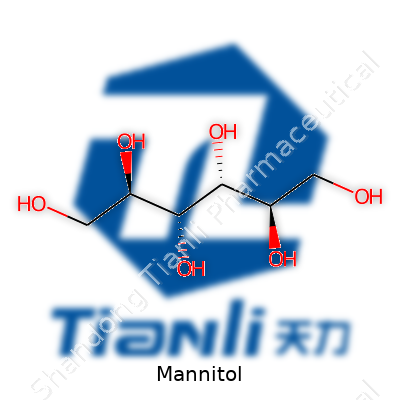
Mannitol doesn’t behave like table sugar. Crystals stay stable even in humid air, resisting clumping and spoilage. A melting point above 165°C means baking it won’t make it breakdown into goo or burn before the rest of the ingredients cook through. Unlike many carbs, it dissolves slowly, giving a cooling sensation as it melts on the tongue. Chemically, its structure consists of six carbon atoms joined in a chain and paired with six hydroxyl groups. That setup keeps it resistant to easy fermentation or breakdown by bacteria, which cuts the risk of dental problems. Its molecular weight clocks in at roughly 182.17 g/mol, and the sweetening power sits next to that of xylitol, but with its own quirks in taste and texture.
Technical Specifications & Labeling
Each batch must clear tight quality checks before hitting store shelves or hospital cabinets. Purity gets measured at over 98%, with tight limits on contaminants like lead or arsenic. Residual solvents also stay under the strict lines that pharmacopoeias lay out. On labels, the name “mannitol” often appears next to its number in the INS system (INS 421) or E number (E421), which helps manufacturers and regulators keep tabs. For pharmaceuticals, labeling highlights the grade, particle size, and endotoxin levels — all factors critical for patient safety. In foods, nutrition panels break down the sugar alcohol content, alerting those on specific diets or with sensitivity to polyols.
Preparation Method
Factories start with starch-rich crops, loading up vats with corn or wheat, then using enzymes or specific acids to break down the complex carbohydrates into simpler sugars. From here, catalytic hydrogenation transforms glucose or fructose into mannitol, using nickel or ruthenium as catalysts under high pressure and controlled temperature. The result comes out as a crystalline solid, which gets washed, purified, and dried. Each refinement step aims to keep unwanted byproducts or trace metals to a bare minimum, ensuring that the mannitol leaves the line suitable for both food and medical uses.
Chemical Reactions & Modifications
Chemists keep tweaking mannitol’s structure to suit specialized tasks. Esterification, where the hydroxyl groups meet acids, leads to derivatives useful in drug delivery. Chemical oxidation turns some hydroxyl groups into aldehydes or acids, opening the door to create other compounds for industry or lab experiments. Mannitol’s performance in reactions rests on its multiple available binding sites, letting it anchor to metals for analytical work or to drugs for controlled release. Some labs modify mannitol’s chain to boost water solubility or tailor its behavior in specific environments, always looking for fresh ways to harness its unique profile.
Synonyms & Product Names
Mannitol travels the world with a suitcase full of names and codes. Shoppers might see “mannite,” “dulcitol,” or “hexanhexol” on ingredient lists, especially on older products or in technical catalogs. Regulatory systems in the EU, US, Japan, and beyond each assign their own codes for traceability and legal compliance, such as E421 or INS 421. Pharmaceutical varieties might go by trade names or manufacturing designations depending on their intended end use and purity. This tangle of synonyms sometimes causes confusion but gives those in the know a way to verify the exact ingredient and specification.
Safety & Operational Standards
Producers follow some of the toughest rules in the business when making mannitol for medicine. Every bag or barrel meant for injection or intravenous drips passes through sterility checks and must show near-zero levels of toxins and pyrogens. Processing plants maintain strict hygiene to keep molds, bacteria, and foreign matter out. In the food industry, mannitol’s status as a “generally recognized as safe” ingredient depends on limits set by the FDA and global agencies. Employees suit up, watch pressure and temperature controls, and document each step for traceability. Large-scale facilities invest in air filtration and round-the-clock monitoring to meet industry standards.
Application Area
Hospitals rely on mannitol during emergencies. Doctors inject it to reduce pressure in the brain after traumatic injuries or before nervous system surgeries. Kidney specialists sometimes use it during dialysis to draw out excess water. Dentists and confectioners use it as a base for sugarless gums, tooth-friendly hard candies, or coatings that stay crispy over time. In diagnostic labs, mannitol works as a calibrating agent or as a carrier in chromatography. Sports nutrition companies mix it into supplements, trusting its stability and mild sweetness. In construction and paints, modified mannitol serves as a stabilizer or bulking agent, showing its utility far beyond the kitchen.
Research & Development
A wave of innovation keeps rolling through mannitol research. Doctors and scientists test its use as a carrier for peptide drugs or proteins, betting that its safety profile and gentle metabolism give sensitive medications a better shot at reaching their targets. Biochemists explore new methods to refine the hydrogenation process, aiming for higher efficiency and less solvent waste. In environmental studies, mannitol’s low reactivity with water and air encourages its use as a safer alternative in industrial processes where reacting byproducts would create pollution. Teams at universities test new derivative compounds, probing mannitol’s many functional groups for possible breakthroughs in drug delivery, food preservation, or even electronics.
Toxicity Research
Every promising product gets held to a high standard in terms of safety, and mannitol’s track record earns broad trust. Standard doses, used under doctor supervision, pass through the body mostly unchanged and exit through the urine. Overconsumption, especially in people with pre-existing digestive troubles, can bring on bloating or diarrhea, a fact that’s plainly acknowledged on food labels across the globe. Toxicological studies in rodents and other lab models report high thresholds for any negative effects, with no evidence of carcinogenic, mutagenic, or reproductive problems. Regulatory agencies in North America, Europe, and Asia keep reviewing these studies and adjust safety guidance as new data come in, looking to keep consumers and patients out of harm’s way.
Future Prospects
Looking into tomorrow, mannitol sits at the crossroads of food science, pharma, and green chemistry. As demand soars for sugar alternatives that don’t spark tooth decay or metabolic trouble, food giants search for new blends and novel sources of this versatile polyol. Researchers in medicine keep testing mannitol as a vehicle to carry fragile molecules safely into the body—hoping to boost treatments for neurological conditions and rare diseases. Sustainability experts dig into crop residue and algae biomass, looking for cheaper, cleaner ways to produce it on a massive scale. Each new method holds promise for cutting waste or making use of materials that would otherwise end up in landfills. With more folks demanding natural ingredients and tested safety in everything from energy bars to life-saving drugs, mannitol’s long career seems ready for another chapter.
Why People in Hospitals Know Its Name
Most folks have never heard of mannitol. In my years volunteering at local clinics, I’ve noticed the word pops up mostly in emergency rooms and hospital notes. Some see mannitol as just another chemical, but in the medical world, it pulls heavy duty. When someone’s brain is swelling after a head injury, or if fluid keeps building up around the brain, doctors often grab mannitol. It helps pull excess water out of the brain and into the bloodstream, where kidneys can filter it out. This approach aims to save lives, not just make numbers look better on a chart.
Getting Rid of Toxins — Fast
When kidneys stop working right, toxins pile up in the blood. Dialysis can help, but sometimes patients still struggle with fluid overload. Here’s where mannitol comes in again. Administered by IV, it spurs the kidneys to send more water into the urine. The process relieves the pressure on the body’s organs and helps patients feel more like themselves. Doctors started using this strategy decades ago, and it’s earned trust through results, not just theory.
Sugar Substitute with a Medical Side
Supermarkets stock mannitol under labels like “sugar substitute” or “low-calorie sweetener.” For people managing diabetes, it means enjoying sweet foods without spiking blood sugar. It’s not just a food trick, though. The body mostly passes it out in urine without digesting it. That’s good news for people looking to keep calories low and blood sugar stable. Food scientists started taking notice of mannitol after seeing how it doesn’t create tooth decay like regular sugar does. For kids and adults fighting cavities, this makes a difference in day-to-day choices.
Medicine’s Helper in Making Pills Work Better
A lot of folks don’t realize that tiny, white tablets they swallow every day rely on mannitol to stay together. Drug makers use it in pills so that the medicine doesn’t turn into a gooey mess or break down too fast. It helps give medicines a stable shape and keeps active ingredients from clumping or separating. If you’ve ever had a chewable tablet, there’s a high chance mannitol contributed to its texture and taste. Pharmacists and manufacturers have seen fewer recalls and complaints because of its consistent results.
Practical Concerns and Cautions
Like any tool in medicine, mannitol can cause trouble if taken without care. People with heart problems or extreme dehydration risk side effects if the dose isn’t right. I remember hearing doctors explain, “Too much fluid out means trouble for the heart.” Medical teams check kidney and heart function first before giving a dose. Research from clinical trials points out the importance of close observation during treatment. The safety track record mainly reflects experiences where trained staff give it under supervised conditions.
Looking Ahead
Demand for mannitol continues to rise—not just in hospitals but in everyday foods and wellness products. Keeping supplies pure and affordable matters. More discussion is happening around balancing access with safe use. Training for caregivers and clear labeling for shoppers go a long way. Every time someone benefits from mannitol—whether fighting off brain swelling or enjoying a sweet snack—they’re relying on decades of real-world experience and scientific checks. That trust didn’t come easy, but it keeps proving its worth.
The Science Behind Mannitol
Mannitol carries a straightforward structure and a clear purpose. This sugar alcohol gets a lot of attention from doctors and pharmacists. They keep reaching for it in high-stakes situations, especially in hospitals. Mannitol travels through the bloodstream, staying mostly unchanged. The kidneys don’t reabsorb it; instead, it pulls water along as it passes through the renal system. This unique ability means mannitol helps the body get rid of extra fluids fast.
Mannitol in Practice: Why Doctors Rely on It
Let’s look at head injuries and certain brain conditions. Emergency departments use mannitol to reduce pressure inside the skull, called intracranial pressure. Swelling in the brain can crush healthy tissue and cut off blood flow. By shifting water from the brain into the bloodstream, mannitol reduces swelling quickly. As a nurse, I remember seeing the relief on faces—both the patient’s and the team’s—when the swelling came down after an infusion.
Mannitol also sees action in kidney protection. Some chemotherapy drugs or dyes used in imaging procedures can stress the kidneys. Doctors may use mannitol beforehand to encourage urine production and flush out toxins, lowering the risk of kidney injury. Studies have shown that this mechanism can make a difference when facing complications that threaten organ health.
Potential Side Effects and Limits
Nothing comes without risks. Mannitol carries its own set of challenges. It draws water out of cells into the blood, but this shift can dehydrate tissues or disturb the balance of salts like sodium and potassium. These changes spark big trouble for people already dealing with heart or kidney problems. Overuse may even trigger heart failure or kidney damage, especially in frail individuals.
Healthcare teams stay alert. Monitoring blood tests before and after mannitol treatments forms a non-negotiable part of the process. Swift action keeps bad side effects from turning into major setbacks. The experience in busy hospital wards shows that careful teamwork makes a real difference.
Why This Matters Beyond the Hospital
Some folks outside the medical field encounter mannitol in their everyday lives. The food and pharmaceutical industries use it as a sweetener in sugar-free products. It doesn’t spike blood sugar, making it useful for people with diabetes. However, large amounts can upset the stomach or cause diarrhea. Real-world experience matches up with research showing that sugar alcohols often don’t agree with everyone’s digestive system.
Looking Ahead: Smarter Use and Better Outcomes
Many medical teams now focus on using mannitol only when truly needed. Greater awareness among doctors and nurses means tighter controls in its administration. Some researchers work on finding alternatives or ways to predict who will benefit without major side effects. Decisions need to come from careful review of the patient’s health history and current condition. Open communication between clinicians and patients is key.
People interested in science or healthcare can learn a lot by looking at how mannitol works inside us. Every time it’s used, professionals weigh the benefits against the risks. That ongoing balance shapes real results for people facing tough odds.
What Is Mannitol and Where Do People Encounter It?
Mannitol gets plenty of attention in medicine and food labs. It’s a sugar alcohol, often used to reduce pressure and fluid in the brain after injuries or as a sweetener for people managing diabetes. Most folks never realize how much planning goes into using a substance like this, yet it’s easy to find—in hospitals, pharmacy shelves, even sugar-free chewing gum.
The Upside Before the Downside
Doctors often reach for mannitol when someone has increased intracranial pressure, or when the kidneys need a nudge to start producing more urine. This can save lives. But using mannitol never feels straightforward. Anytime you use something that changes the body’s water and salt balance, there’s a tradeoff.
Poking at the Big Side Effects
The main things that worry folks who’ve actually given or received mannitol are dehydration and electrolyte imbalances. Mannitol pulls water out of tissues—pretty helpful after a brain injury. But sometimes it does the job too well. People start losing more water than they can drink back, and then sodium or potassium levels start to dance around unpredictably.
Even in the controlled setting of a hospital, big shifts in electrolytes can knock the heart or brain off balance. Nurses and doctors keep a close eye on the patient’s blood chemistry after each dose, since confusion, muscle cramps, fast heartbeats, or seizures can creep up. Some patients throw up, get headaches, or even feel chest discomfort, so having skilled clinicians around isn’t just a precaution—it’s a must.
Less Talked-About Risks
For some patients, especially those with tough kidneys, mannitol builds up in the system. Fluid can start to pool in unexpected places, leading to swelling or even heart problems. I’ve seen cases where kidneys just stopped cooperating after mannitol made them work overtime. People with underlying heart failure or kidney disease need an extra layer of caution.
Allergic reactions can happen too, though that’s much less common. Some people show up with rashes or tight chests, rarely full-blown anaphylaxis. In the hospital, this means switching drugs on the fly, which is never simple for someone already in a fragile state.
The Human Side of Treatment Choices
No medication should be used on autopilot. Before giving mannitol, clinicians weigh out not just the benefits but the real risk of side effects kicking in. I’ve watched experienced residents debate the timing and dose, knowing that for every patient helped, there’s another who winds up in a deeper tangle of medical problems. We can’t brush aside what happens to folks who get dehydrated, who end up sicker than before.
Regular monitoring, stepped-up lab testing, and honest conversations with patients and families turn out to be the actual solution. Better training about who gets mannitol and when it’s safer to skip it can make a difference. Technology like point-of-care blood tests, smarter IV pumps, and good old-fashioned team huddles often catch trouble before it spirals.
The Bottom Line: Thoughtful Medicine
Using mannitol is a reminder that in medicine, each choice sends ripples through the whole body. No clear-cut answer fits every situation. The smartest thing is to spot warning signs fast, talk with everyone involved, and not let routine overpower careful thinking. Whether you’re a patient or someone holding the chart, remembering the real-world effects of drugs like mannitol steers care in the right direction.
Understanding Mannitol and Its Uses
Mannitol plays a big role in hospitals, especially for patients who need quick relief from swelling or high pressure in the brain. Doctors count on this sugar alcohol because it draws water out of tissues, helping reduce pressure fast. You’ll most often hear about it in emergency rooms or during surgeries, not in a family medicine clinic.
How Mannitol Gets Into the Body
Mannitol reaches the bloodstream through an intravenous (IV) route. In other words, it’s put straight into the veins, skipping the stomach and digestive system entirely. This isn’t by accident. The digestive tract doesn’t absorb mannitol well when swallowed as a pill or powder. The IV route works much quicker and has a more predictable effect. If a brain injury or high eye pressure threatens damage, doctors can’t afford to wait for a pill to slowly kick in.
Why IV Is the Go-To Method
Hospital staff mix mannitol with sterile water, forming a clear solution that goes into a bag. Nurses connect it to an IV line, usually in the arm. From there, gravity or a pump helps the medicine flow right into the bloodstream. This approach means the medical team can control the dose almost minute by minute. If a patient shows signs of improvement or starts having side effects, they adjust the drip speed or halt it altogether. No waiting for digestion; no guessing if a swallowed tablet did its job.
People dealing with conditions like glaucoma or certain types of kidney failure sometimes get mannitol, too. For folks with kidney issues, mannitol acts as a diuretic, sparking urine production. Still, doctors don’t just hand it out lightly. It’ll be used only when the kidneys have enough function to handle the boost, so the extra water doesn’t end up causing new problems.
Safety Factors That Need Attention
Mannitol isn’t for home use. The hospital setting allows for close monitoring. Electrolytes, fluid balance, heart rate—all these get watched. I remember a patient who got mannitol for head trauma. The team checked labs every hour to make sure sodium and potassium didn’t swing out of range. Mannitol may pull water out of swollen areas, but it can also trigger dehydration or strain the heart in older folks. The IV team keeps an eye on the injection site, checking for redness or leaks. Flushing the IV line with saline helps keep everything clear for future medications.
Looking Ahead: Smarter, Safer Use
Doctors and pharmacists keep studying mannitol’s right dose and the safest infusion rates. Technology now helps with accurate electronic drug pumps. Clear labeling, barcoding, and double-checking meds before they go in the bag cut down on mix-ups. In some places, newer osmotic agents are under review, but mannitol remains a trusted option for quick, targeted relief. Teaching teams and patients about risks—like fluid overload, kidney stress, or allergic reactions—bolsters safety too.
Mannitol's story is one of fast response and precise care. Its use is measured, intentional, and rooted in science. It brings hope in emergencies, but always under the sharp eyes of the medical team trained to use it wisely.
Looking Beyond the Label
Mannitol stands as a staple in many emergency rooms and intensive care units. Doctors often turn to it to pull fluid out of brain tissue or lower pressure in the eyes. It sounds pretty straightforward, but the real story starts before mannitol ever touches a vein. From personal time in the pharmacy and collaborating with ER teams during high-risk moments, I’ve seen cases where mannitol did more harm than good. So, the trick with mannitol is less about using it and more about knowing who just can’t handle it—or needs some serious precautions.
Kidney Concerns
Mannitol pulls water with it as it travels through the body, working as an osmotic diuretic. If the kidneys already struggle—let’s say from acute kidney injury or long-term kidney disease—mannitol can tip the scale the wrong way. In patients with kidney problems, mannitol sometimes sticks around and builds up. That can draw even more water out of cells, flooding the body with excess fluid, and, in rare situations, cause heart failure. Natural to want a fix for high brain pressure, but pushing mannitol into the bloodstream of someone with weak kidneys can quickly backfire.
Heart Problems and Fluid Balance
Fluid overload isn’t just a kidney story. Anyone with heart failure lives with a delicate balance of salt and water. Mannitol drags fluid into the blood before kidneys can remove it, and that extra load can swamp a weak heart. Each time I talk with a cardiac nurse or see heart failure patients being re-admitted after aggressive diuretic use, I’m reminded how easy it is to tip the balance.
Electrolyte Shifts Are Real
One thing that never gets enough attention is how fast electrolyte levels can shift. Mannitol can trigger big swings in sodium or potassium. Low sodium can cause seizures or confusion. Too much potassium makes the heart rhythm unstable. Labs aren’t just a checkbox—they guide every step of the way. During ICU shifts, team members keep a close eye on labs before every bag of mannitol. A small oversight opens the door to big complications.
Dehydration and Blood Volume
Infusing mannitol into someone who’s already dehydrated can dig a deeper hole. When cells lose water too quickly, the blood gets thicker, blood pressure falls, and organs suffer. A patient treated for stroke who arrives dehydrated may end up far worse if mannitol goes in unchecked.
Head Injuries—Not Always a Green Light
Sometimes, physicians look to mannitol right after a head trauma or brain surgery. But not every swollen brain calls for it. If bleeding in the brain is ongoing or blood pressure drops too low, mannitol’s fluid-shifting magic can stop helping and start hurting. Some of the most tragic outcomes I’ve seen have involved well-meaning attempts to lower brain pressure without fully understanding the underlying issue.
Practical Safeguards
Every specialist, from ER docs to pharmacists, agrees: patient selection matters more than the medication. Checking kidney function, heart health, and fluid status isn’t just a protocol—it’s a lifeline. Monitoring urine output, blood pressure, and lab values protects patients from the hidden dangers mannitol brings. Corticosteroids, hypertonic saline, or mechanical measures sometimes replace mannitol in high-risk cases.
No one solution fits everyone. Sometimes, pushing pause and asking for a consult makes more of a difference than any bag of medication on the shelf.