Urea: From Discovery to Future Prospects
Historical Development
Long before factories churned out fertilizer by the trainload, urea’s story began in the early 19th century. Chemist Friedrich Wöhler’s 1828 experiment showed that an organic compound could come from inorganic sources — a moment that marked more than a breakthrough in the lab. Wöhler’s synthesis from silver isocyanate and ammonium chloride wasn’t just good science; it knocked down walls between living chemistry and lifeless minerals. This simple molecule, CH4N2O, later shaped the backbone of the world’s agricultural systems. Large-scale manufacture didn’t take off until the Bosch-Meiser process paired cheap ammonia with state-supported industry. Every pile of urea on a farm today stands on the foundation built by chemists who believed complex life processes could be unraveled and repeated.
Product Overview
Urea shows up in the farm fields, chemical plants, medical labs, and even daily hygiene products. Packed as dense white granules or in crystalline powder, it drains smoothly through storage bins and behaves well in mixing machines. Nitrogen content lands at 46%, more than any other solid fertilizer, which appeals to everyone squeezed by rising costs. Its broad presence isn’t only about abundance — it’s the fit for use, handling, and long shelf life. In medicine, it leverages the body’s own biochemistry, filtering into creams for dermatology, diuretics, and diagnostics, showing a flexibility many industrial chemicals only wish they could match.
Physical and Chemical Properties
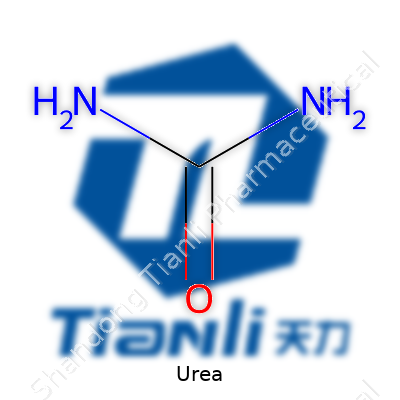
Urea crystals glint with a white hue, melting in a steady way near 133°C. Poorly soluble in ether but free-mixing with water and alcohol, urea fits well in both watery liquid blends and solid-state mixes. No smell, little taste, harmless to touch, so gear stays simple. Laid out as (NH2)2CO, the molecule carries two amine groups flanking a carbonyl, and this shape welcomes bonding with a variety of chemicals. Urea’s amide group takes a starring role in forming hydrogen bonds—crucial for stability in blends and compatibility with volumes of other substances.
Technical Specifications and Labeling
Shelf space in warehouses benefits from clear standards and honest reporting. Reliable labeling includes total nitrogen content, biuret levels, moisture percentages, and granule size. Food and agriculture regulators like the FAO, EFSA, and China's GB set the exact rules, not just for numbers but also for allowed contaminants and heavy metals. Producers run samples through consistent protocols, tracking heavy metal residues, dusting potential, and caking traits. Practical details such as packaging type, recommended storage, and precautions against moisture invasion are required on every bag and drum, channeling information straight to operators rather than hiding behind technical jargon.
Preparation Methods
Fertilizer plants use ammonia and carbon dioxide as the starting blocks, both coming from natural gas. Under heat and high pressure, these react to create ammonium carbamate, which dehydrates into urea. Not much goes to waste; heat recovery and gas recycling are baked in at most plants, cutting costs and curbing emissions. Purification strips away byproducts and water, leaving behind granules or prills ready for packing. Scale and stewardship of these steps mean less resource strain and greater affordability for the rural economies depending on steady deliveries.
Chemical Reactions and Modifications
Urea reacts kindly with acids to form salts, and teams up with aldehydes to form resins—the kind of chemistry that sets adhesives, lacquers, and plywood glue. Mixed with formaldehyde, urea resins create hard finishes for flooring, chips for particleboard, or durable tableware. It breaks down into ammonia and carbon dioxide in soil under farmer’s feet, feeding crops as long as temperature and moisture allow. Urea’s backbone also bends for pharmaceutical intermediates, cleaning solutions, animal feed supplements, and even as a defrosting aid for runways. The bond flexibility in its amide groups lets it serve as a raw material for dozens of industries outside fertilizer.
Synonyms and Product Names
Depending on the application, urea answers to names like carbamide, carbonyl diamide, isourea, or sometimes just by its trade labels—Azotin, Carbamide Resin, IsoUrea. The world market rarely sticks to a single name or formulation, so regional synonyms pop up in trade paperwork and scientific writing. For example, Europe’s “carbamide” marks pharmaceutical grade, while fertilizer markets use “urea” or “prilled urea.” Understanding these shifts avoids confusion in contracts, safety sheets, and cross-border shipments.
Safety and Operational Standards
Delivering urea from factory to field means following regulations that stretch from cargo labeling to aquifer protection. Workers rarely need heavy gear for handling dry urea since the substance itself shows low health risk at normal exposure. Dust masks and gloves prevent throat or skin dryness with prolonged contact, but field exposure stays low. Regulatory ceilings keep biuret, a byproduct that stunts sensitive crops, below certain limits. Warehouses require dry, cool conditions, safe stacking, and regular checks so urea stays free-flowing. Shipping codes include HAZMAT registration for large volumes, not due to acute toxicity but for proper traceability, especially as downstream products hit medical and food industries.
Application Area
Crop fields soak up most of the world’s urea, with over 150 million tonnes spread each year to keep wheat, rice, and maize green and tall. Livestock operations pick up urea for non-protein nitrogen feed, giving ruminants cheap protein alternatives. Industrial use claims a smaller but important share, pushing urea-formaldehyde resin production, diesel exhaust cleaning (AdBlue/DEF fluid), and glue production for wood processing. Dermatology embraces it for skin hydration, and the pharmaceutical sector employs it in diuretic drugs and laboratory reagents. Every market relies on the same baseline qualities: high nitrogen, clean handling, and predictable behavior in process lines and use.
Research and Development
Teams in labs focus on tweaks to improve both environmental impact and farm productivity. Slow-release coatings on granules, for instance, help prevent nitrogen leaching and save farmers money over the long haul. Biotechnology researchers look at enzymes that break down urea in the soil, with hopes of matching release rates to plant demand. Upstream, process engineers stress recycling CO2 and maximizing ammonia yield from renewable resources. Digital modeling now predicts greenhouse gas emissions at field scale, using real application data. This intersection of on-ground practicality and leading-edge technology reflects the priority placed on stewardship and sustainability.
Toxicity Research
Direct urea exposure at typical usage rates brings little cause for concern to humans or livestock, though researchers keep heads up for potential risks from misuse or accidental spills. Drinking water regulations in regions like the EU and US set limits for urea breakdown products, keeping nitrite and nitrate within safe zones for infants and sensitive ecosystems. Overapplication to fields risks groundwater pollution, and aquatic life in rivers near fertilizer runoff shows stress with long-term nitrogen overload. Chronic exposure studies dig into hormone disruption risks associated with high urea derivatives in urine, but routine clinical and agricultural uses stay well under danger thresholds.
Future Prospects
As the planet faces tighter land and water constraints, the world expects more from urea each passing year. Future production lines could shift to green ammonia, derived from electrolysis-driven hydrogen, slicing the carbon footprint deeper than fossil fuel-fed routes. Smarter coatings and precision application tools mean less runoff and pollution, driving efficiencies up and input costs down. Regulatory pressure will likely widen as researchers document subtler impacts on soil microflora and neighboring aquatic ecosystems. Alternative sources of nitrogen and the intersection with plant genetics might shake up the current dominance of urea, but the legacy of high-yield, adaptable chemistry will keep it a mainstay for decades to come.
Behind Every Green Field: The Role of Urea in Food Production
Walk through any thriving field of wheat, corn, or rice, and you'll see what urea can do for a harvest. Most people don’t realize how much urea matters for growing food, even though it rarely makes headlines. Farmers often mix urea into the soil to help crops stretch tall and strong. Urea packs a lot of nitrogen, which plants need for leafy growth. The green color you see on healthy plants often comes straight from nitrogen. Without enough, crops stall and yields drop, hurting both farmers' incomes and our food supply.
Global food security ties to fertilizer availability, especially over the last few years. The COVID-19 pandemic, war in Ukraine, and shipping disruptions all made it tougher or pricier to get urea in certain countries. As a result, some places faced higher grocery bills or even empty shelves. The world’s population keeps rising. Modern farming must squeeze more from less land, so urea isn’t just something for big industrial operations — it keeps food affordable for everyone.
Industrial Workhorse and Everyday Helper
Fertilizer grabs most of the headlines, but urea's not a one-trick pony. Factories use urea to help make resins for plywood, particleboard, and other building materials. If you’ve lived in a house with wood furniture or floors, urea’s likely played a part in making that happen. Urea formaldehyde resins stick wood particles together, shaping the cheap and sturdy boards found at hardware stores. Without it, builders would pay a lot more for raw materials, and home prices might creep even higher.
Beyond construction, urea keeps trucks and cars running cleaner. Modern diesel vehicles need to cut down nitrogen oxide emissions, so they use a fluid called Diesel Exhaust Fluid (DEF). This fluid comes straight from urea mixed with water. Truckers and logistics companies have depended on DEF to stick to emissions regulations. Skimping here means more smog in city air and eye-watering fines for transport companies. Clean air is not just a talking point for politicians or scientists — everyone breathes it in.
Health Care and Clean Water
Doctors and nurses use urea-based creams to help skin stay hydrated. People with dry or cracked skin, especially those with diabetes or eczema, rely on these creams for daily comfort. In my own family, a relative has used prescription urea ointment for years to manage cracked feet, especially in winter. It’s not magic, but it makes a difference you feel by the end of a day.
Wastewater treatment plants rely on urea, too. It acts as a key chemical to remove pollutants and control odors before water returns to rivers or lakes. If municipal water treatment falls short, it doesn’t take long for neighborhoods to notice bad smells or even risk to health.
Looking Ahead: Risks and Smarter Use
Relying so much on urea puts the world in a tricky spot. Overusing nitrogen fertilizer leads to runoff, where rain carries extra nitrogen into rivers and lakes, setting off algal blooms that choke aquatic life. Fields keep producing, but down the line, fish die off and water tastes strange. Farmers and researchers need to try smarter ways to use urea, including timing applications and mixing with other techniques, to cut back on wastage. Digital tech, soil sensors, and educational programs point toward a future where farmers get more from every dollar spent on urea and less of it goes to waste.
Rising prices, export bans, and unpredictable supply all push everyone — growers, builders, drivers, and everyday consumers — to think about where their goods really come from. Digging into urea means facing tough choices, but it also means every plate of food and gallon of clean water carries a story that stretches from the farm, to the factory, and right back to our own homes.
What Urea Actually Does
Walk down a drugstore aisle and plenty of lotions shout about moisture, healing, and healing dry skin. Flip any of those tubes around—"urea" often pops up on the ingredients list. Urea, a compound our bodies naturally make, lands in a lot of creams aimed at seriously parched or rough skin. Dermatologists point to its ability to draw in water and soften up rough patches. At lower strengths, urea acts like a sponge, soaking up water from deeper layers and pulling it to the outer skin. At higher concentrations, urea takes apart the stuff holding dead skin cells together, smoothing out calluses and cracked heels. Both effects matter for anyone who battles rough, dry skin or lives where winters suck all the moisture out of your face and hands.
Real-World Uses and Benefits
Doctors have recommended urea ointments for decades, especially for eczema or thick, tough calluses. Even people with diabetes—who face a higher risk of skin breaking down—use urea-based creams to avoid painful cracks and open wounds. Studies back up these uses. A German study found that 10% urea lotions helped eczema patients keep their skin stable and smooth, with fewer steroid creams needed. More research supports mixing urea with other drugs (like clotrimazole for athlete’s foot), which smooths the skin and lets medicine do its job better.
Inside My Bathroom Cabinet
I started reaching for urea cream a few years ago for flaking hands after too much hand-washing during the pandemic. The relief didn’t come overnight, but I noticed the difference after a few days—a gritty palm finally turned smooth. Even for scaly elbows and the thickened heels from daily walks, nothing so far competes with a cheap urea cream from the pharmacy. Some friends with eczema or even psoriasis swear it’s the only over-the-counter stuff that helps when everything else leaves burning or sticky residue.
Safety Concerns and What Actually Happens on Skin
Consumers sometimes worry about sticking something called "urea" on their face, maybe because the term brings up images of urine. But the ingredient in creams gets manufactured in labs, without any funky byproducts. Most people tolerate urea well, though a sting or a mild itch can happen if the skin’s already raw. Stronger concentrations—think 30% instead of 5%—sometimes burn if skin has open cuts. Allergic reactions stay rare, much less than some fragrances or preservatives found in fancier creams. If you’ve got very sensitive skin or use stronger acids, patch-testing a bit behind the ear or elbow helps dodge rashes or stings.
Who Should Be Careful?
While urea soothes many skin woes, a few people may want extra advice. If someone’s skin has large open sores, or if a rash is spreading, it helps to ask a doctor before slathering on strong urea formulations. Parents using urea cream for young kids—especially those with eczema—should look for lower percentages and avoid the eyes and lips. Pregnant or breastfeeding women don’t face special risk with urea applied on skin, according to medical reviews.
Looking at Alternatives and Solutions
For cracked or scaling skin, plain petroleum jelly or a lanolin cream works if allergies crop up. Glycerin-based lotions can boost hydration too, but may feel sticky or greasy. People allergic to fragrance usually find urea creams cause no trouble because most skip perfumes. If drugstore creams fail, a doctor’s visit makes sense. Prescription urea formulas run stronger, sometimes blended with steroid creams for those who need serious skin repair.
Putting the Facts First
Urea has earned its place in the skin care world by delivering smoother, softer results for countless people living with rough skin, eczema, or cracked feet. I’ve seen it help my own hands and many others get back to normal, especially in harsh or dry climates. The science—mixed with straightforward experience from doctors and users—shows that urea works well and poses little risk to most people. Choosing a reputable product, reading the label, and starting with a small patch clears up most worries for anyone curious about trying urea.
The Chemical Path From Air and Gas to White Granules
Urea sounds simple. Most people see it as tiny white pellets scattered over fields. The journey from raw material to fertilizer tells a story about chemistry, industry, and the world’s food supply. Anyone who’s walked through farmland might have smelled the sharp ammonia scent that sometimes wafts from freshly fertilized ground. That scent connects back to urea’s origins.
Starting With Ammonia and Carbon Dioxide
It all starts with natural gas. Factories extract hydrogen from methane found in natural gas. Air provides nitrogen. Together, these two elements form ammonia through the classic Haber-Bosch process—a breakthrough that dates back over a century. This reaction literally feeds billions because ammonia builds the backbone for making urea. With hydrogen and nitrogen combined, the next step adds carbon dioxide—often captured from the same process that creates ammonia.
Bringing the Elements Together
Inside a reactor vessel, ammonia meets carbon dioxide at high temperatures and pressures. These extreme conditions push the two chemicals to react, and urea forms along with water as a by-product. The factory’s challenge is separating urea from everything else. Workers cycle the mixture through stages, concentrating the urea into a thick solution and allowing water to evaporate off. In big facilities, these steps run around the clock, watched closely by technicians trained to manage both the chemistry and the powerful equipment involved.
From Solution to Pellet
Once concentrated into a thick liquid, urea takes on a solid form. Factories spray the solution through nozzles at the top of tall towers. As droplets fall, they cool and harden into round granules. These pellets pile up at the bottom, ready to be bagged and shipped. Some places might form urea as prills or in other shapes, but the end product always keeps nitrogen in a form crops can use. Granulated urea handles easily, resists caking, and dissolves steadily in soil.
Why This Matters
Synthetic urea revolutionized global agriculture. Before its invention, farmers dug up guano or rotated crops endlessly to avoid depleting soil. Today, about half of the world’s food grown depends on nitrogen fertilizer. China, India, Russia, and the United States run enormous urea plants that anchor their food systems. While organic sources of fertilizer remain in use, they simply don’t supply enough nitrogen to meet the demands of modern crop yields.
Environmental and Economic Costs
Behind these white granules, signs of strain on climate appear. Urea production demands natural gas and releases greenhouse gases. Runoff pollutes rivers and coastal waters—dead zones where only algae grow. Better designs for fertilizer application and modern factory scrubbers help, but the balance gets trickier every year. I’ve seen farmers test slow-release urea coatings to cut losses and keep more nitrogen in the field, and researchers search for new catalysts to make ammonia at lower temperatures. The industry faces a puzzle: feed people, but not at the planet’s expense.
Looking Forward
Every bag of urea holds a choice. Society needs to keep people fed, yet waste and emissions threaten water, air, and future harvests. Smaller tweaks, like precise application and plant science breakthroughs, help stretch each granule’s benefit. I walked through fields that used both synthetic and manure-based fertilizer, and the difference in crop vigor showed plainly. Still, sustainable farming asks for tougher decisions upstream, not just in the field. Drawing nitrogen from the air and carbon from waste streams—without burning so much fuel—may change how the world makes food for the next generation.
The Reality Behind the Storage
Walk into any farm supply store and you’ll find bags of urea stacked and waiting. Some bags seem fresh, others a bit weathered. This common sight brings up a real question from folks who know the value of every dollar they spend on fertilizer: Does urea actually expire, or does it just sit there, ready to boost the next crop season?
How Long Can Urea Last?
Anyone who’s spent a season or two in farming has heard all kinds of stories about fertilizer gone bad. My own experience tells me that urea holds up much longer than many worrywarts let on. Urea isn’t like milk or bread—it’s a solid chemical with a surprisingly steady makeup. If kept away from moisture, it can stay usable for years. Some chemical producers even suggest a shelf life up to two decades, but in an ordinary farm shed, five to ten years is a solid figure. Moisture remains the big threat. If water sneaks into those bags, urea clumps up, breaks down to ammonia, and the product loses its punch.
Storage Matters Most
A dry, well-ventilated space determines if urea outlasts seasons or falters after a single rainy month. I remember seeing a stack in a friend’s barn—left on bare concrete, bags sagging, some torn open. Those bottom bags absorbed enough moisture to turn into hard cakes. It doesn’t take a chemist to spot the problem: anytime urea cakes, it’s probably lost a good chunk of its strength. Folks say dry plastic totes or sealed drums work wonders. Keep bags up on pallets, clear off the ground, away from barn doors that catch the driving rain.
Why Shelf Life Actually Matters
Working fields means guessing the weather, juggling planting schedules, and hoping nothing goes sideways. Some years, half the nitrogen application ends up stashed away when storms roll in. Nobody wants to toss leftover urea if next spring’s budget already feels tight. There’s nothing wrong with keeping urea on hand, but only if it’ll still feed the crop right. Studies, including research by major agrochemical organizations, show that a ton of urea stored properly keeps its nitrogen content season after season. Eventual caking or yellow color signals trouble. Any crusted or liquified product means the fertilizer probably lost more than half its value.
What Can Farmers Do?
Too much ruined urea adds up over a career—missed yields, wasted money. Farmers have found pretty direct answers. Invest time into storing fertilizer properly. Cover, pallet, seal up. Check moisture barriers. Broken bags invite more than just clumping. Local extension offices sometimes offer on-the-ground advice or recommend moisture meters, which remove the guesswork. Some farms set up yearly rotation, using older stocks first and buying only enough for a season or two. These small habits deliver real savings and keep fertilizer working hard for the land, not leaking value.
Better Storage, Better Results
All things considered, urea outlasts worry—if treated right. Respect for a bag of white prills rivals respect for rain in late spring. Handling and storage matter more than the printed “best before” date, especially on a working budget. Quality fertilizer rewards good habits, and the land remembers which farmers look after their tools.
Looking at Urea in the Field
Urea stands out in agriculture for its high nitrogen content and low cost. Farmers turn to it for everything from corn to rice. It dissolves quickly in water and gets moving in the soil right after application. Plenty of folks ask if it makes sense to mix urea with other fertilizers. The short answer: sometimes. The truth is, a bit more digging is needed before dumping different bags in the same spreader.
How Urea Reacts With Others
Mixing fertilizers isn’t just shuffling powders or granules. Some blends play well together, but others may clump, lose nutrients, or wind up wasting money. With urea, moisture is the main troublemaker. It grabs water from the air, and that can turn a dry mix into a headache. I’ve seen clumps form so tight that they wouldn’t even break apart in a spreader. Adding ammonium nitrate to urea can help in cool climates, but it can turn sticky in areas with high humidity.
Urea and potassium chloride usually don’t fight much, but when folks mix urea with ammonium sulfate, a chemical reaction can release ammonia. That turns precious nitrogen into a gas, and profits start floating off into the air. Blending urea with phosphates brings another issue. The two react and start caking if moisture sneaks in. Farmers then find themselves with a solid brick instead of a useful blend.
Losses Hit the Pocketbook
Nutrient losses can be huge. Research has shown that mixing urea and superphosphate can bump up nitrogen loss by as much as 30 percent if spread together in warm, damp conditions. Every bag lost equals a smaller harvest and a tougher year. I still remember watching a neighbor’s wheat respond poorly after a bad mix; he learned the hard way that shortcuts in the shed cost big in the long run.
Managing Blending For Better Results
A few smart steps protect yields. Storing urea and other fertilizers apart in dry sheds stops caking and chemical reactions before they even start. Blending urea with potash for same-day use works for many, but it should be spread right after mixing. Keeping things dry is key. If storage humidity creeps above 60 percent, granules turn pasty and stop flowing. Manufacturers sometimes add coatings to slow water uptake, but that comes with extra cost.
Custom blends pay off most when folks talk directly with crop advisers or agronomists. Specialists check for chemical compatibility and recommend what pairs make sense for the soil, weather, and crop plan. The results usually beat the guesswork of mixing on the fly.
Smarter Fertilizer Use Builds Healthier Land
Blending doesn’t just affect nutrients. Poor mixes waste money and leave behind hotspots of chemicals that burn roots or cause groundwater trouble. Sticking to recommended guidelines lets every dollar in the fertilizer budget pull more weight. A soil test, matched with a good plan, keeps nutrients balanced and the farm’s future steady. Years of experience remind me that the best results never come from shortcuts—only from those extra moments spent learning and double-checking before the spreader heads out to the field.